Stem Cells: Overview, Differentiation Potential, and Use of MSCs in Bone Grafting
The use of stem cells in bone grafting applications continues to grow in popularity. This is due in part to aging patient populations that require more advanced materials for successful surgical outcomes. In parallel, surgeons are seeking more advanced technologies to treat these patients.
Stem cells are unique because they can be stimulated to change, or “differentiate,” into bone cells by their environment. Some advanced synthetic bone graft materials even have the ability to stimulate stem cells to become bone cells. But what exactly is meant by the broad term “stem cells” and what benefits do they promise? This blog covers an introduction to the biology of stem cells and how stem cells may be used specifically for bone regeneration.
Introduction to Stem Cells
Although their clinical use remains controversial, embryonic stem cells have amazing power to morph into virtually any organ or tissue under specific chemical and physical conditions. These cells are called “pluripotent,” from the Latin root pluri- meaning many. Other kinds of stem cells, called “multipotent” or “adult stem cells,” are more differentiated, which means they can form into a more limited variety of tissues. For bone regeneration applications, adult stem cells can either be autologous stem cells (coming from the patient who is being treated, also known as autograft) or as an allograft, i.e., allogenic stem cells (coming from another human, usually a cadaver). Naturally, any bone graft product containing donated allogenic stem cells, i.e., cellular allografts, has to undergo a number of FDA regulations and protocols to ensure its safety and asses the efficacy of use.
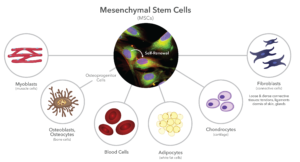
One type of adult stem cell commonly associated with bone grafting applications is a mesenchymal stem cell, or MSC. These cells were first identified in the mid–1970s. MSCs are partially differentiated, so they are capable of forming a variety of structural or stromal tissues, such as bone, cartilage, tendons, muscles, blood, and fat (Figure 1). They are limited to these tissue types because they are embryologically derived from the mesoderm, hence the term ”mesenchymal.” In contrast, the ectoderm is responsible for forming skin and the central nervous system, while the endoderm is responsible for the functional cells of major internal organs, such as the liver, pancreas, and kidneys.
MSCs are found throughout the body, particularly the pericytes nested within the structural components of microvascular walls. Therefore, bone marrow is especially rich in MSCs. MSCs are spindle shaped but not particularly unique in their morphology. Because they readily replicate, they grow on tissue culture plates into “colony forming units,” or CFUs. They are best chemically identified by specific antigens, called “Clusters of Differentiation,” in their cell membranes, such as CD105, and CD44. MSCs are rare, representing less than 0.1% of the nucleated cells in the body, and they further decrease with the age of the patient. Although MSCs can replicate more than 50 times, their doubling rate is slow, more than 1-3 days, and decreases with advancing age.
Allogenic MSCs are capable of being used in patients because they are thought to be immune privileged. This means they do not contain major HLA (Human Leucocyte Antigens) antigenic components in their cell membranes that the patient recipient sees as foreign. Therefore, they are not detected by the patient’s immune system when implanted. By contrast, HLA compatibility is required for any organ transplant to prevent rejection. MSCs can therefore theoretically be used to treat any patient without the concern for histocompatibility.
Mesenchymal Stem Cell Use in Bone Grafting
Bone is one of the most easily regenerated structural tissues, so it is no surprise that MSCs are readily available in the bone marrow. Hence, bone marrow aspirate (which is harvested from cancellous bone) is widely used in combination with other bone graft materials. Due to the popularity of bone marrow derived stem cells, tissue banks eventually figured out a way to process allograft bone while keeping the donor MSCs alive. Today, both mesenchymal stem cells (MSCs) and more differentiated stem cells called osteoprogenitor cells (OPCs) (Figure 1) are harvested from cadaver bone. These MSCs and OPCs are then added to DBM (demineralized bone matrix) and/or cancellous bone chips from the same donor to create a variety of allograft stem cell products.
There is a lot of debate regarding the efficacy of transplanted MSCs and OPCs. It is not clear whether these cells actually survive, much less divide into viable cells, after transplantation. However, there is data to support that they act as paracrine cells (signaling cells) by releasing cytokines and other growth factors into their environment. Thus, they indirectly stimulate bone regeneration, rather than to directly differentiate into osteoblasts.
The Future of Stem Cells in Bone Regeneration
Modern processing techniques have greatly expanded access to stem cells for use in surgery. For procedures that require bone grafting, it is expected that surgeons will continue to explore the use of mesenchymal stem cells, osteoprogenitor cells, and related technologies to grow new bone and help patients heal more fully. While stem cell use remains a topic of debate, these cells hold great promise in the field of bone regeneration and will likely continue to play a role in future bone regeneration technologies.
Image sources:
- File is licensed under the CC Attribution-Share Alike 4.0 International license. Original image available at: https://www.researchgate.net/figure/Mesenchymal-stem-cells-stained-with-fluorescent-dyes-2-Nucleus-are-in-blue_fig5_278827757.
As a recognized leader in advanced bone graft technologies, Biogennix is committed to bringing high-quality educational content to our field. This blog will cover technical topics ranging from basic bone graft science to advanced osteobiologic principles. We’ll also discuss market trends and industry challenges. We thank you for reading and invite you to learn more about us here.
