Bone Composition: Part 2 – Bone Matrix
As discussed in the previous post on the Bone Composition blog series, there are three primary components of bone composition: cells, organic bone matrix, and inorganic bone matrix. In Part 1, we talked about bone cells and how they play a crucial role in bone formation and remodeling. In this blog, we will focus on bone matrix and its organic and inorganic components.
Bone Matrix Composition
Bone matrix is a musculoskeletal tissue that consists of a complex composition of organic and inorganic components. Although bone matrix composition can vary amongst individuals, it typically has the following composition (Bostrom, 2000*):
- Inorganic component: bone mineral – 60-70%
- Organic component: proteins (e.g., collagen & growth factors), lipids, and other macromolecules – 22-35%
- Water – 5-8%
Due to this multi-component composition, bone matrix serves a variety of biological functions. As the main component of the skeletal system, the collagen and mineral components of bone matrix provide biomechanical support for the entire body and protect vital organs. Bone mineral additionally serves as a reservoir for calcium and phosphorus, key elements used throughout the body. Bone matrix also contains non-collagenous proteins such as growth factors that are actively involved in bone formation, repair, and remodeling.
Organic Bone Matrix – Proteins, Lipids, and Other Macromolecules
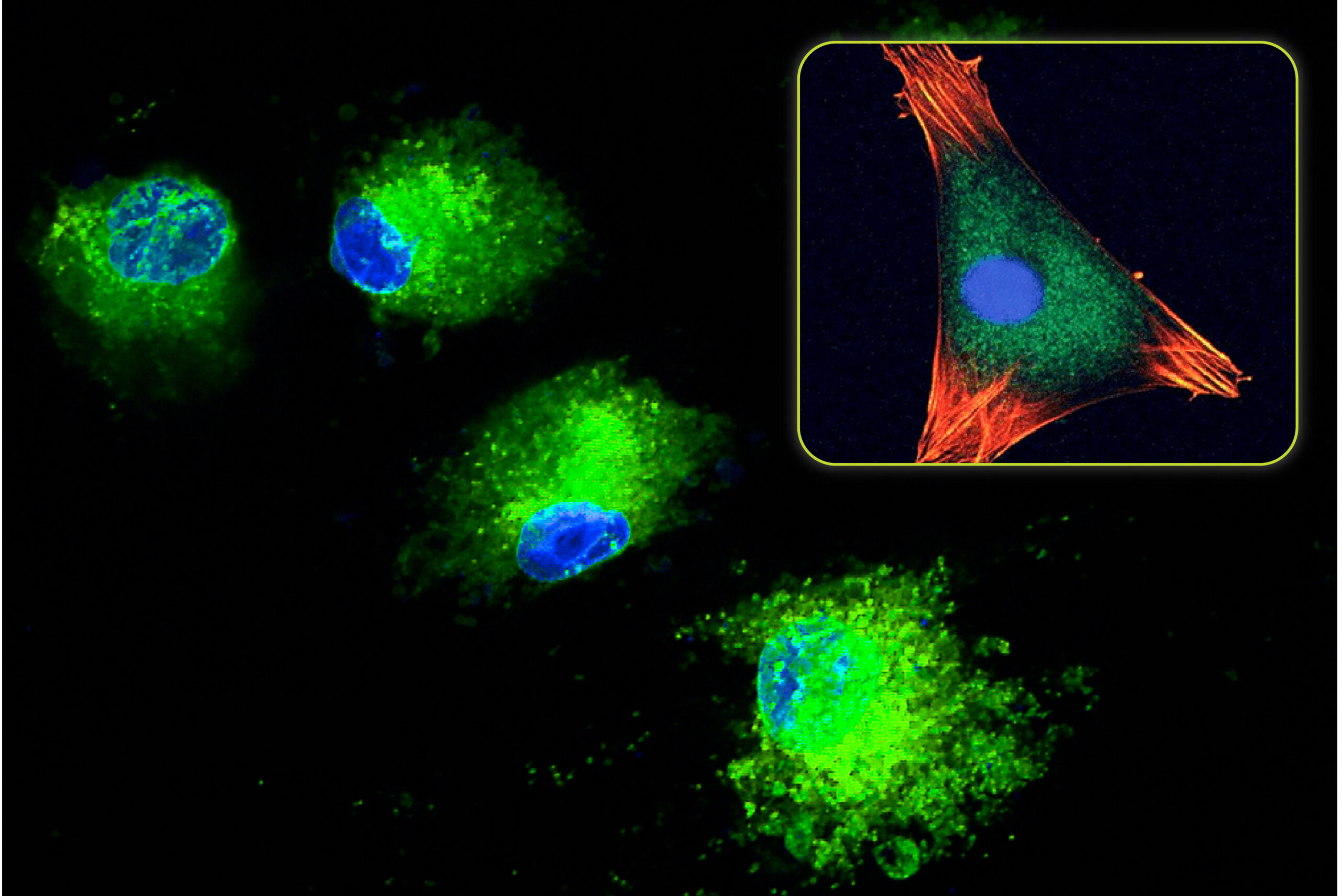
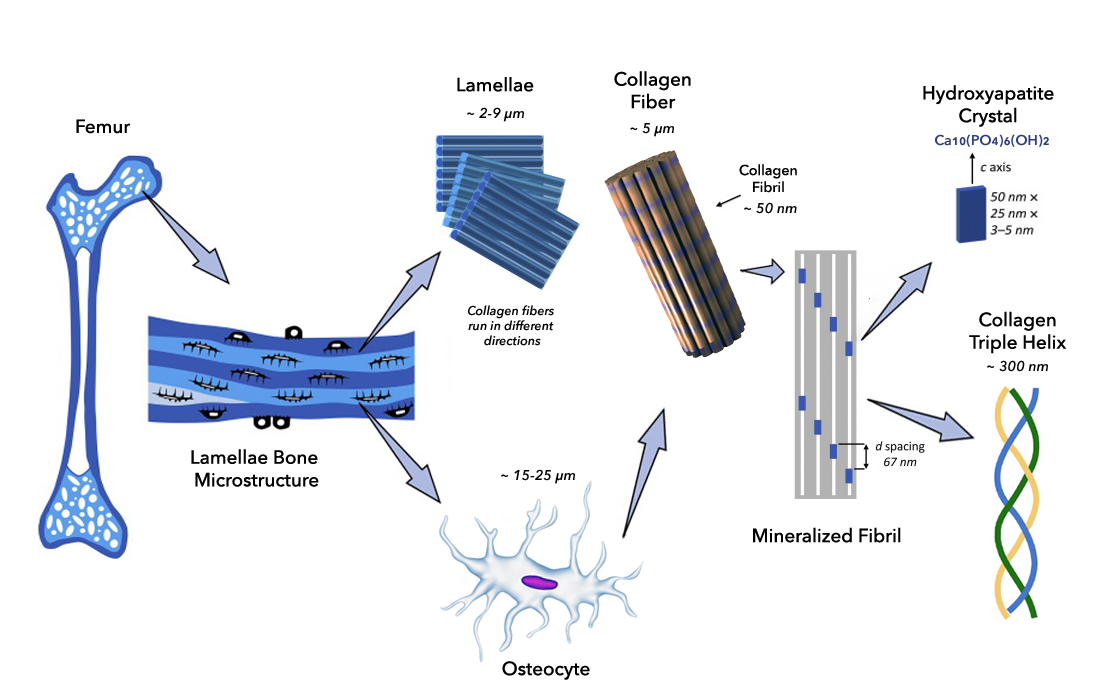
Collagen is the main protein component in bone accounting for approximately 90% of the organic bone matrix composition. By convention, the collagen that is found in bone is referred to as Type I collagen. However, there are more than 20 types of collagen found in various tissues throughout the body. They all have common properties, such as being high in certain amino acids (e.g., glycine and hydroxyproline), and are formed into a triple helix of approximately 300 amino acids for each chain, and 300nm long. After secretion by cells, these large triple helices aggregate together into collagen fibrils. The collagen fibrils are held together by inter-molecular and inter-fibrillar bonds, or cross-links.

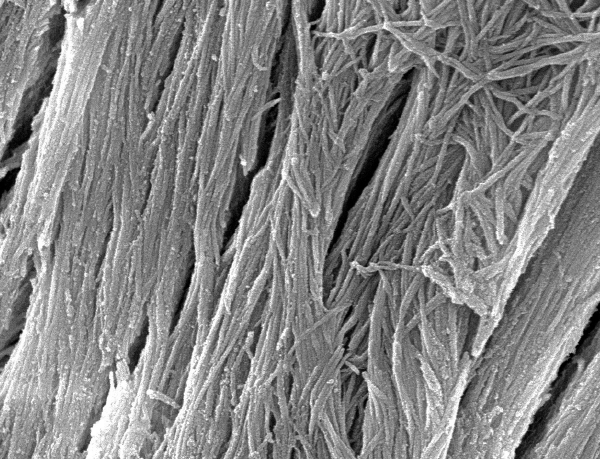
The collagen fibrils then combine together in precisely aligned bundles, leaving tiny (65nm) spacings which cause bands in the fibrils (Figure 1). The bundles of microscopic fibrils then aggregate into even larger collagen fibers (Figure 2).
This hierarchical arrangement gives collagen fibers an incredibly high tensile strength. This makes the bone matrix composition very strong but flexible at the same time. The spacing also provides a seeding location for the formation and crystallization of bone mineral elements, such as calcium and phosphorous. The combination of the collagen and the mineral form the structural basis of the bone matrix.
In addition to collagen, other proteins are also found in bone. One class of proteins called growth factors are specialized proteins that directly control cellular healing processes throughout the body. In bone, a variety of growth factors are involved in the bone formation process including (Note: Some listed growth factors represent a group of similar proteins, e.g., BMP-1 through BMP-14):
- Bone morphogenetic protein (BMP)
- Transforming growth factor-β (TGF-β)
- Platelet derived growth factor (PDGF)
- Insulin-like growth factors (IGF)
- Fibroblast growth factor (FGF)
- Vascular endothelial growth factor (VEGF)
Due to their involvement in bone formation, these proteins are present when mineralized bone matrix is being formed. As a result, the growth factors become trapped in the mineral phase. In response to an injury, these growth factors can be re-accessed by osteoclasts as the mineral phase is being resorbed and remodeled.
Aside from collagen and growth factors, bone also contains other macromolecules such as lipids, carbohydrates, and other non-collagenous proteins. Found in smaller quantities, these components aid in maintaining normal bone turnover (resorption and remodeling), assist with normal bone metabolism, and interact with cells in the bone marrow.
Inorganic Bone Matrix – Bone Mineral
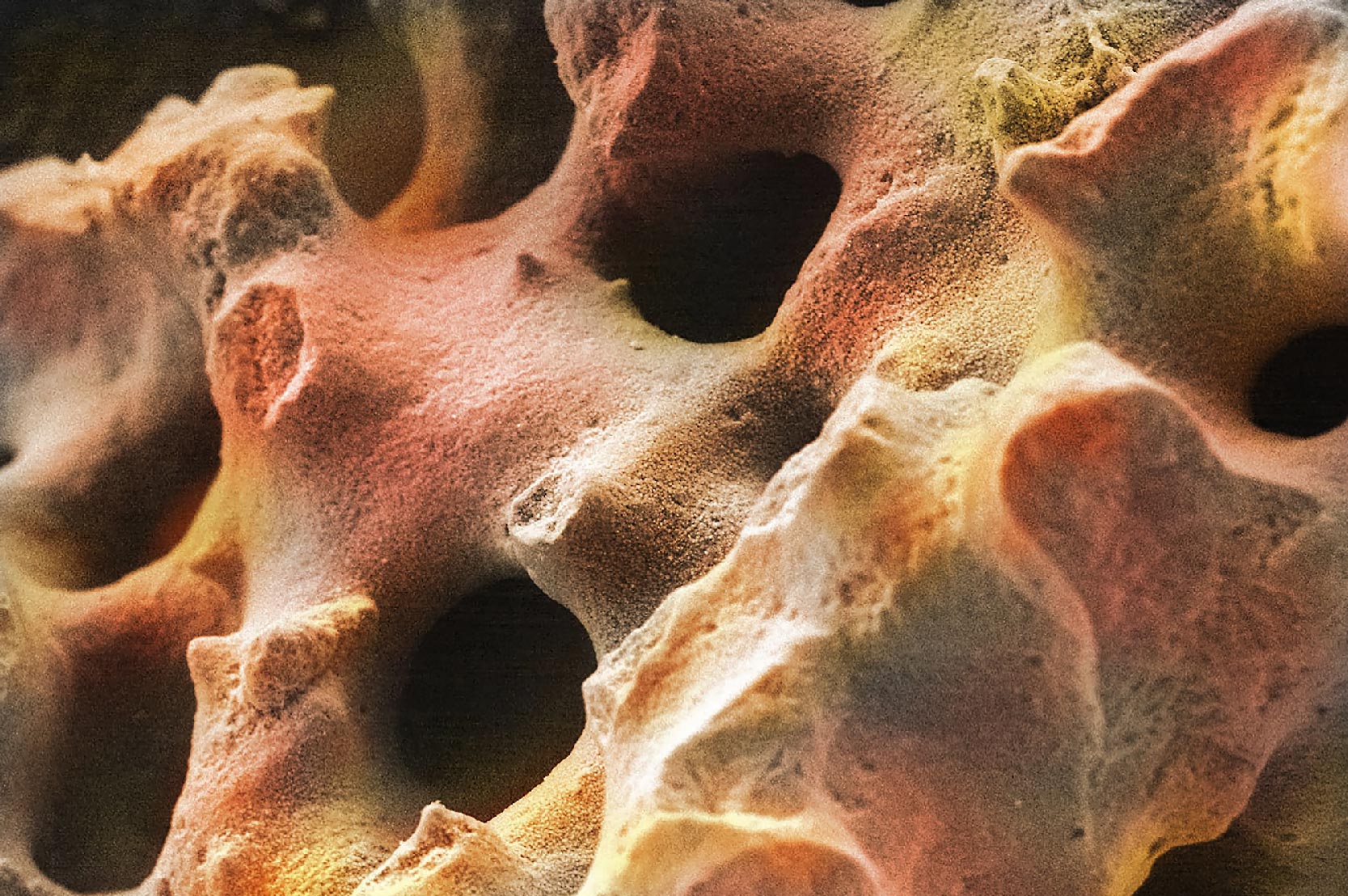
In most medical school textbooks, the mineral in bone is commonly referred to as hydroxyapatite. Often these books will also state that the hydroxyapatite is formed outside of the cell due to the precise molecular structure of collagen fibers. Although true, this is a simplification of very important and complicated biological processes.
When mineralized bone is formed, the bone mineral components in the interstitial fluid precipitate, or form, “hydroxyapatite” (HA) crystals (Figure 3). This initially occurs within the tiny nano-gaps between the collagen molecules. This calcification process progresses over time to fully mineralize the bone. The bone mineral crystals that are formed have rod or plate shapes measuring 5-20nm in size. Although pure hydroxyapatite has a very specific formula [Ca10 (PO4)6 (OH)2], the HA mineral in bone is extensively “substituted” with other ions existing in the body fluids. For example, magnesium (Mg), manganese (Mn), and strontium (Sr) ions are often exchanged for calcium (Ca). Additionally, carbonate (CO3) and chloride (Cl) ions are often substituted for either phosphate (PO4) or hydroxyl (OH) groups. These bone mineral substitutions tend to increase the solubility of bone, making it easier for osteoclasts to break it down. In contrast, fluoride is often added to drinking water to decrease the solubility of bone, especially to preserve teeth enamel.
Carbonate is especially complicated and important because up to 10% is also incorporated into bone as calcium carbonate. It is also substituted either in the phosphate or hydroxyl sites. Therefore, bone mineral components are most accurately stated as either hydroxycarbano-apatite (HCA) or carbonated hydroxyapatite (CHA).
In bone, calcium phosphate and calcium carbonate work together to form hydroxyapatite (HA) crystals. These hydroxyapatite crystals use collagen fibers as a scaffold and, once properly adhered, give bone its rigidity and hardness. Collagen fibers give the bone its flexibility and motion range. In other words, the organic component of the bone matrix helps resist tension whereas the inorganic component helps resist compression.
Bone Composition: Putting It All Together
In this two-part blog series, we have learned that bone has three main components: cells, an organic bone matrix component, and an inorganic bone matrix component. These three unique parts make bone a true composite material that combines advantages of multiple materials to create something better than the individual parts. The cellular component is crucial for bone formation and remodeling. The organic bone matrix component provides tensile strength from the collagen fibers, and biological activity and homeostasis from the growth factors. The inorganic bone matrix components are essential for compressive strength and serve as a large reservoir of calcium and phosphorus. All three component are necessary for bone to function properly and maintain its amazing ability to heal and repair itself, allowing organisms like humans to live healthy, active lives.
References:
* Bostrom Metal et al. “Chapter 13. Form and Function of Bone. Orthopaedic Basic Science: Biology and Biomechanics of the Musculoskeletal System.” Buckwalter J et al. Rosemont, IL: American Academy of Orthopaedic Surgeons: pp. 320-369 (2000).
Image sources:
- File is licensed under the CC Attribution-Share Alike 4.0 International license. Overlaying text removed. Original image available at: https://commons.wikimedia.org/wiki/File:Collagen_fibrils_in_rabbit_skin.jpg
- “SEM deproteined bone – wistar rat” by Bertazzo S. File is licensed under the CC Attribution-Share Alike 3.0 Unported license. Overlaying text removed. Original image available at: https://commons.wikimedia.org/w/index.php?curid=20904735
- File is licensed under the CC Attribution-Share Alike 4.0 International license. Substantial text alternations, resizing, repositioning, and image replacements were made. Original image available at: https://www.sciencedirect.com/science/article/pii/S174270611830672X
As a recognized leader in advanced bone graft technologies, Biogennix is committed to bringing high-quality educational content to our field. This blog will cover technical topics ranging from basic bone graft science to advanced osteobiologic principles. We’ll also discuss market trends and industry challenges. We thank you for reading and invite you to learn more about us here.