The Importance of Biomimetic Porosity in Bone Formation: Part 2 – Porosity-Strength Relationship
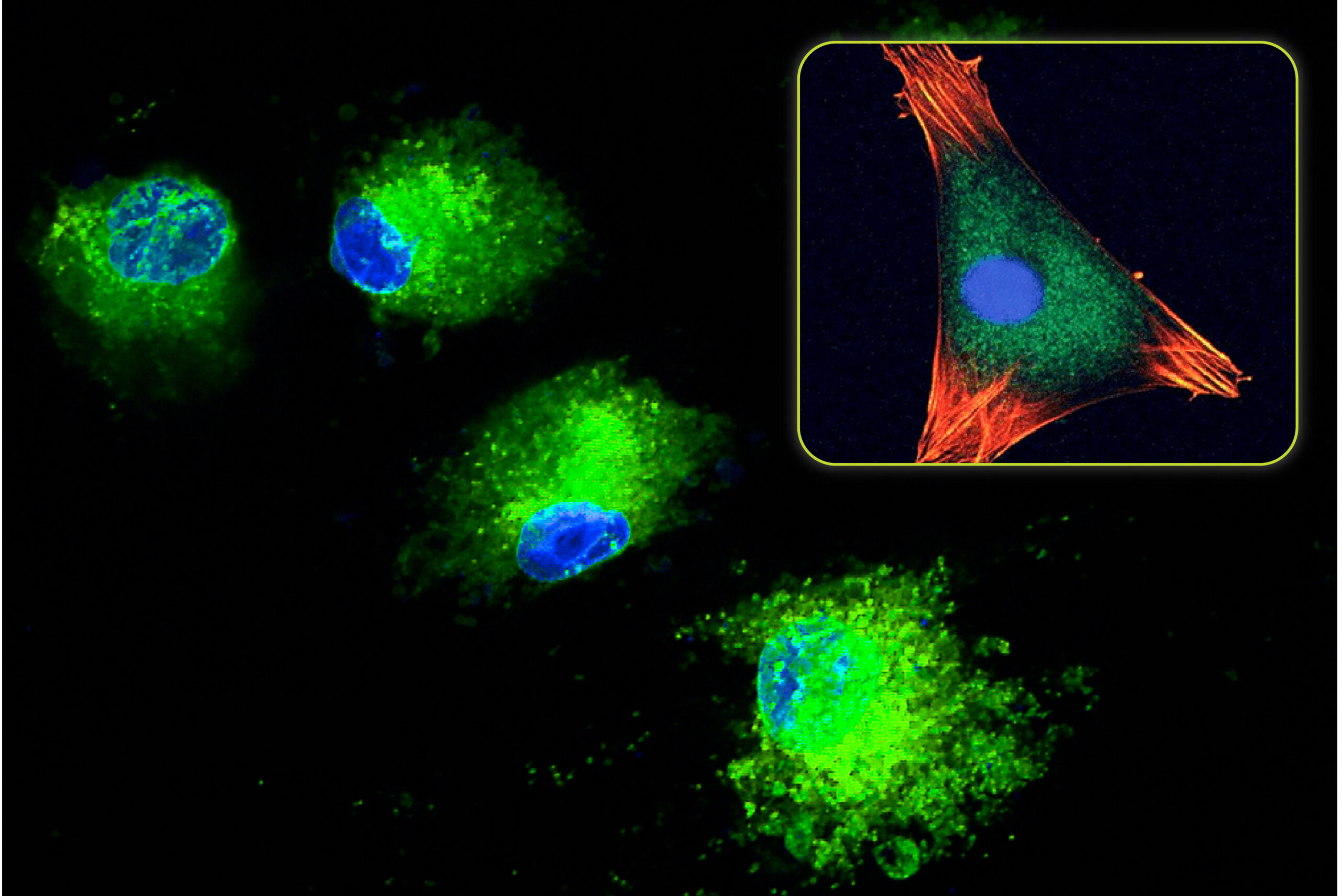
All bone grafts need to function as a scaffold for new bone formation and support bone growth on their surface. This property is called osteoconductivity and is a critical characteristic for a successful patient outcome. Osteoconduction is a complex process that is primarily affected by a material’s composition and porosity. Porosity, in particular, can be modified to improve a material’s osteoconductive ability.
In part one of this two-part blog, we explored the role of pore architecture in bone formation, including the effects of pore size, interconnectivity, pore volume, and surface area. Porous graft materials are osteoconductive, in part, because their structure provides an abundance of surface area for cells to infiltrate and adhere. However, higher porosity materials aren’t always superior, as increasing levels of porosity lead to lower graft strength. In clinical practice, lower graft strength is problematic, because surgeons usually need to manipulate the graft material before and during implantation. Overly porous and brittle bone graft scaffolds may get crushed into a powder, eliminating some of their osteoconductivity. As a result, finding a graft material with the right balance of strength and biomimetic porosity for bone formation is key when it comes to getting the best clinical outcomes. Let’s investigate this dynamic in more detail.
The Porosity-Strength Tradeoff

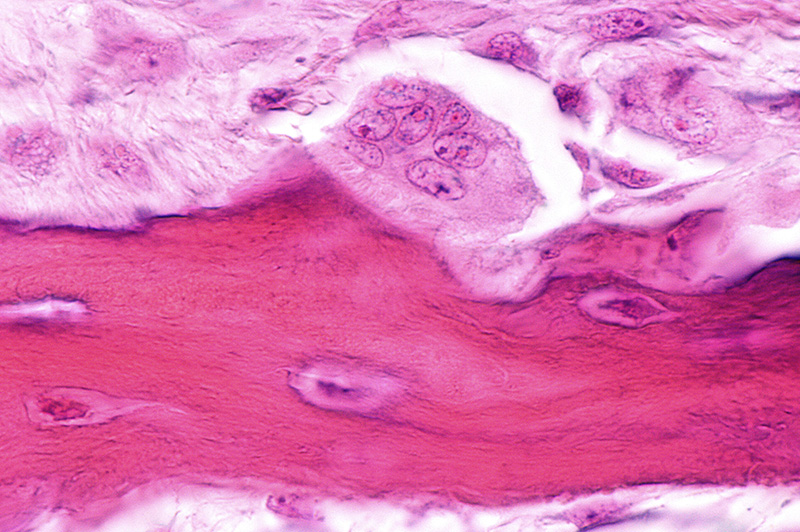
Typically the ability of a material to support bone growth can be increased with the addition of porosity (assuming proper pore sizing and interconnectivity). A graft material in a solid, non-porous form would be very strong and difficult to damage while handling. However, upon implantation there would be no way for cells to grow into the material, dramatically slowing or preventing the new bone formation process. Bone formation would be solely limited to the material surface and would be dependent on resorption to create more room for new bone. By adding porosity, the area for bone formation increases, but less material is present and the structure is weakened. As such, bone graft porosity and strength have an inverse relationship. With less material, the structure has less strength compared to a solid block. As porosity continues to increase, the strength continues to decrease. With a highly porous graft material, there is more room for bone ingrowth, but the structure has a low density, is more brittle, and is more prone to intraoperative crushing than less porous materials. These characteristics are due to the diminished mechanical strength resulting from the increase in void volume (i.e. porosity). Conversely, dense graft materials with smaller pore volumes are more durable, but there is less room for bone in-growth, reducing their osteoconductivity.
Bone Graft Manufacturing and Properties Affecting Strength
Material strength can be evaluated according to several metrics, including compressive strength (crush strength), tensile strength (elongation strength), and fracture strength (toughness). For bone graft materials, the compressive strength and its resistance to crushing are the key properties. In the production of synthetic bone graft materials, graft manufacturers use a variety of different techniques to create porosity. This includes:
- Foaming Techniques – gases used to create bubbles and spacing within a material
- Sintering – heating powders or particles to a temperature just below the melting point; causes the material to bond at the contact points; spacing between the particles functions as the porosity
- Sacrificial Void Creation – embedding a temporary particulate material (such as wax spheres) in the main bone graft material; then dissolving or burning off the temporary material to create a void
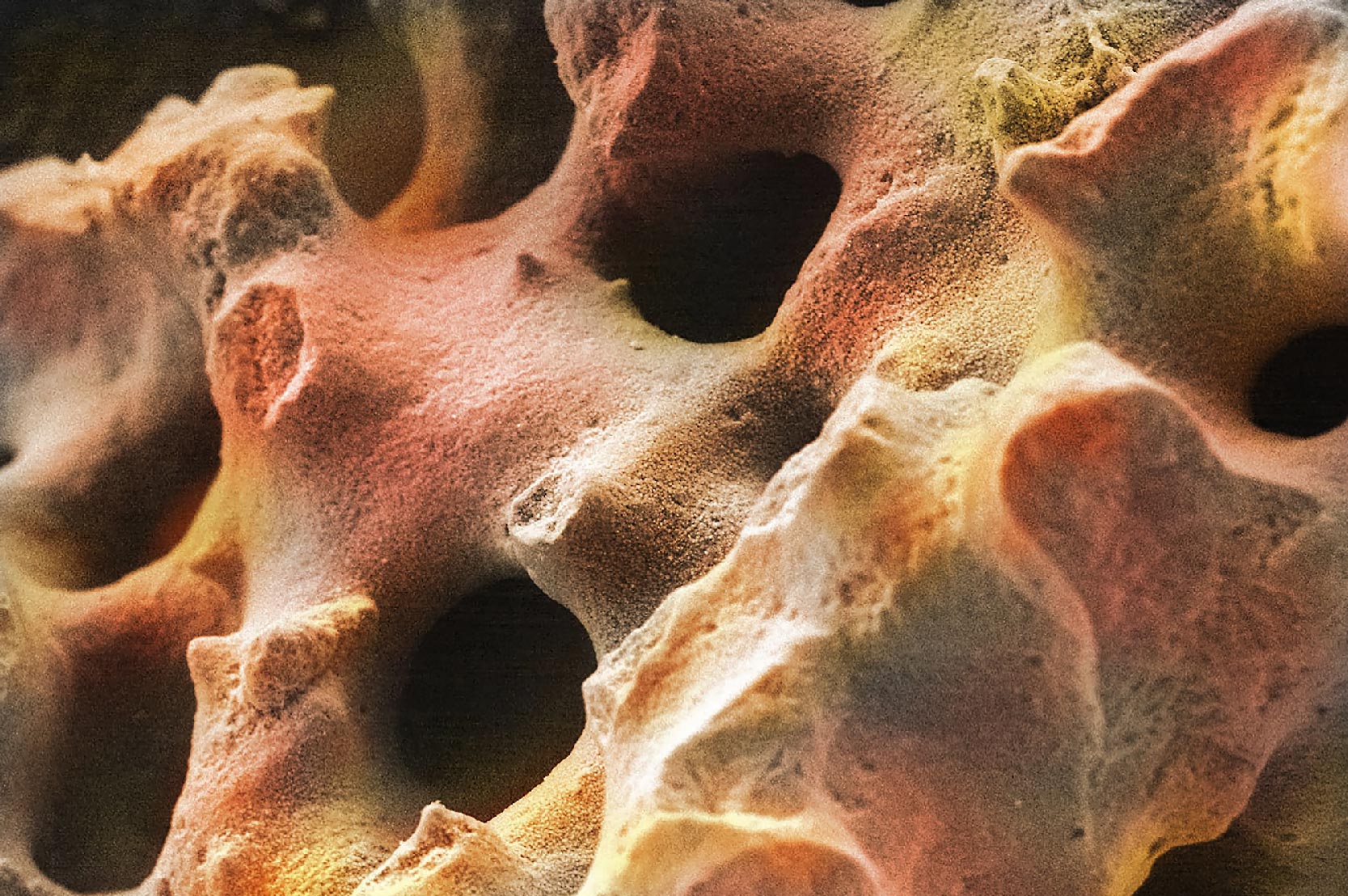
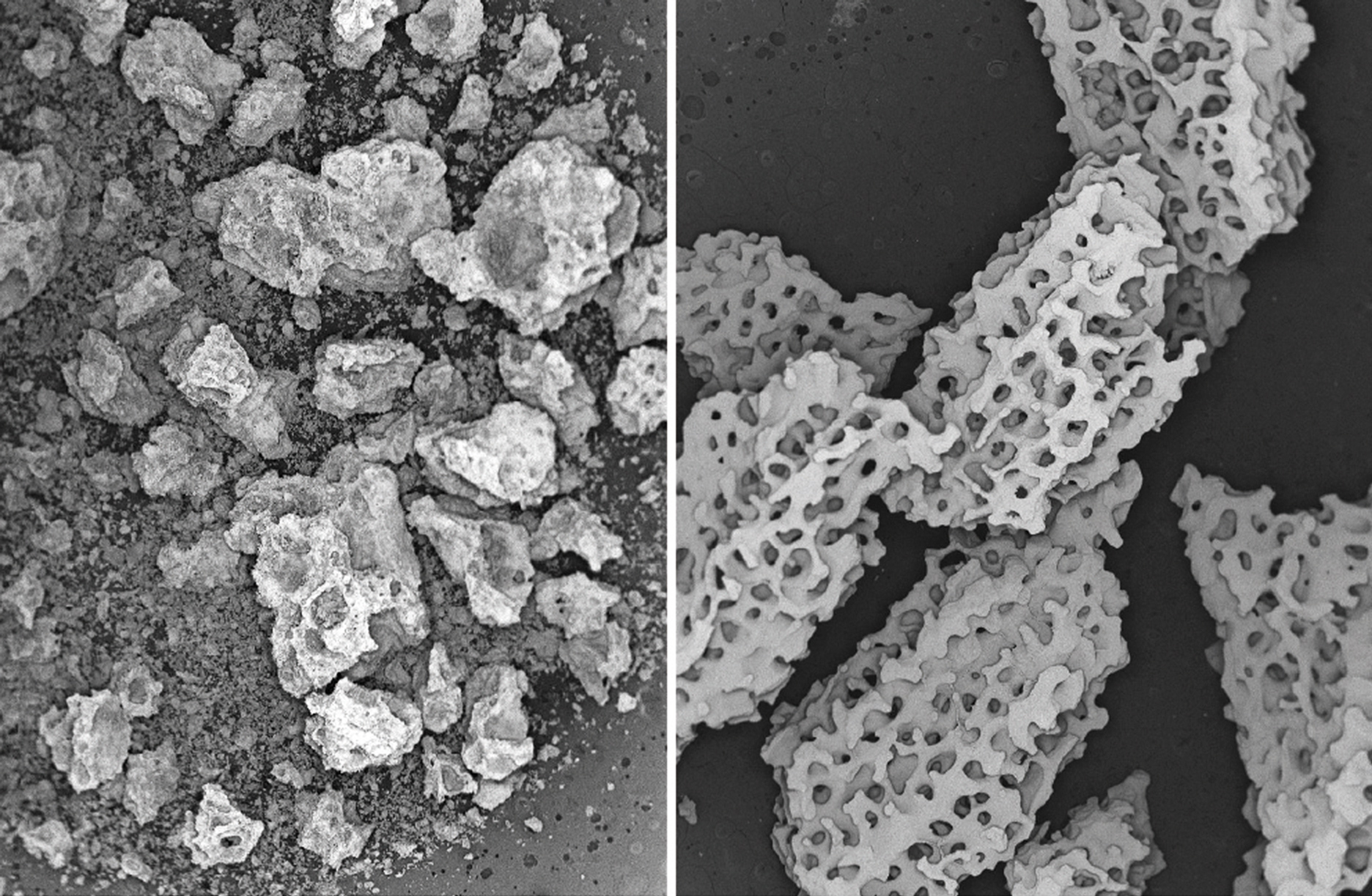
The use of these bone graft manufacturing techniques allows the formation of porous materials. However, the structures are characterized by non-uniform porosity with a broad size range and irregular geometry that is easy to fracture or compact during handling. In addition, these standard manufacturing methods can result in porosity without true interconnections which are critical for neovascularization and osteoconduction. In particular, grafts with an abundance of pores too small for bone-ingrowth (< 100 microns) are weakened by pores that do not provide any osteoconductive benefit (Hing 2005). Additionally, grafts with sharp or irregularly shaped pores are more prone to crushing or fracturing than rounded surfaces. The sharp corners/edges in these porous materials act a stress point where fractures are easily initiated. This makes these materials more prone to breakage.
Conversely, grafts containing rounded and arched pore surfaces have reduced stress points and a higher fracture resistance. These types of structures have an interconnected porosity and architecture that is similar to human bone. This biomimetic property describes bone graft materials that mimic the natural structure of cancellous bone and arguably provide the ideal scaffold for new bone growth.
Why a Non-Structural Bone Graft Needs Strength
Compressive strength is important for bone grafts even though they are not intended to be load-bearing implants. Indeed, their primary function is to facilitate bone formation. This means that they need to be strong enough to survive surgical handling with an intact porosity. In a typical bone graft procedure, surgeons may manipulate graft material by:
- Kneading it into the appropriate shape
- Blending and mixing it with autograft
- Packing it into implant sites, manually or with tools, to ensure close contact with native bone
- Compressing and extruding it with surgical hardware like MIS graft funnels
These manipulations subject the graft to substantial compressive and shearing mechanical forces that can easily crush overly porous, brittle materials. As a result, finding the right balance between strength and biomimetic porosity for bone formation is an important consideration for surgeons.
Biomimetic Porosity: Optimal Bone Formation and Graft Strength

Due to the inverse relationship of strength and porosity, it is important to find an optimal middle ground.
This would be a material that has enough porosity for an effective bone formation response and enough strength to survive the implantation process. Nature has shown that cancellous bone is such a structure. Graft materials with biomimetic porosity are ideal for bone formation because they are designed to provide an optimal combination of porosity and strength. Biomimetic graft materials have pores that have a similar geometry to cancellous bone, with rounded openings and a fully interconnected architecture. Furthermore, their porosity and consistent size is optimal for bone ingrowth, rather than being too large or too small. Biomimetic porosity also leads to a stronger graft material, which makes it able to survive implantation and be a more effective osteoconductive scaffold.
While many synthetic graft materials lack fully biomimetic properties, bone grafts containing TrelCor® technology feature a fully biomimetic pore architecture that encourages the fast bone ingrowth while providing optimal material strength that can sustain intraoperative handling. In contrast to other synthetic graft materials that are overly porous and brittle, biomimetic materials can maintain their porosity, and thus their osteoconductivity, even after aggressive manipulation and shaping by surgeons.
As a recognized leader in advanced bone graft technologies, Biogennix is committed to bringing high-quality educational content to our field. This blog will cover technical topics ranging from basic bone graft science to advanced osteobiologic principles. We’ll also discuss market trends and industry challenges. We thank you for reading and invite you to learn more about us here.