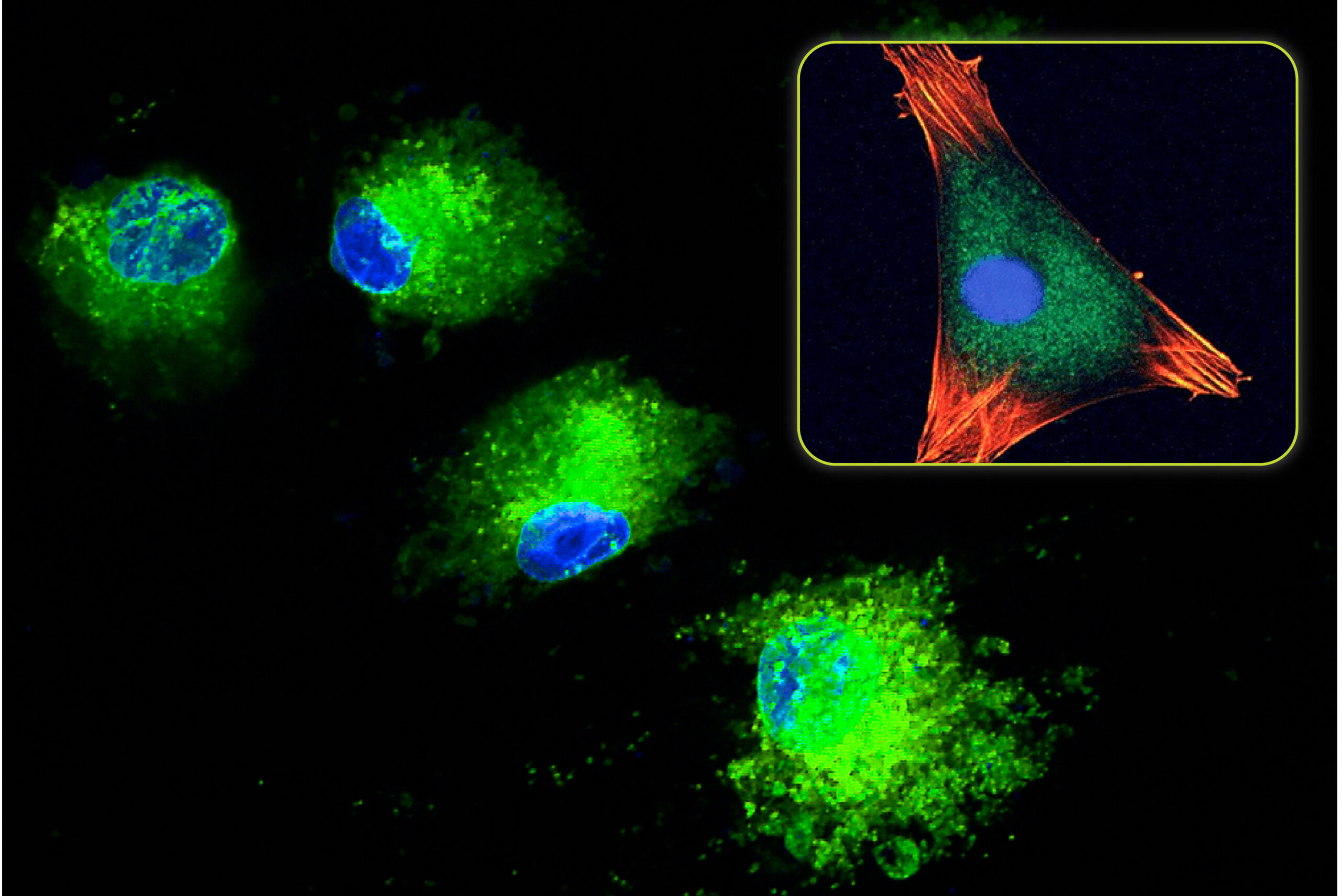
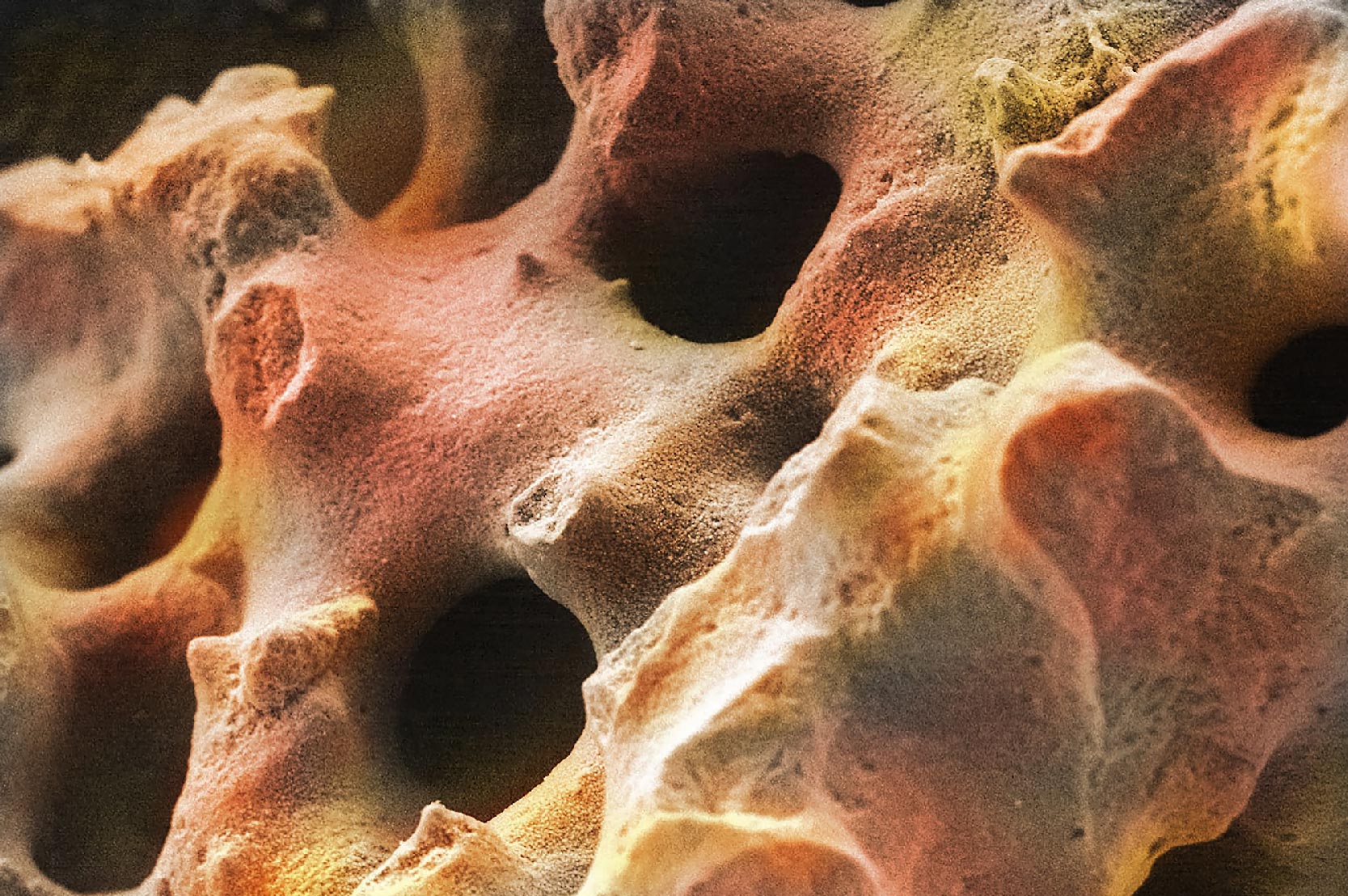
The Importance of Biomimetic Porosity in Bone Formation: Part 1 – Pore Size, Interconnectivity, Volume, and Surface Area
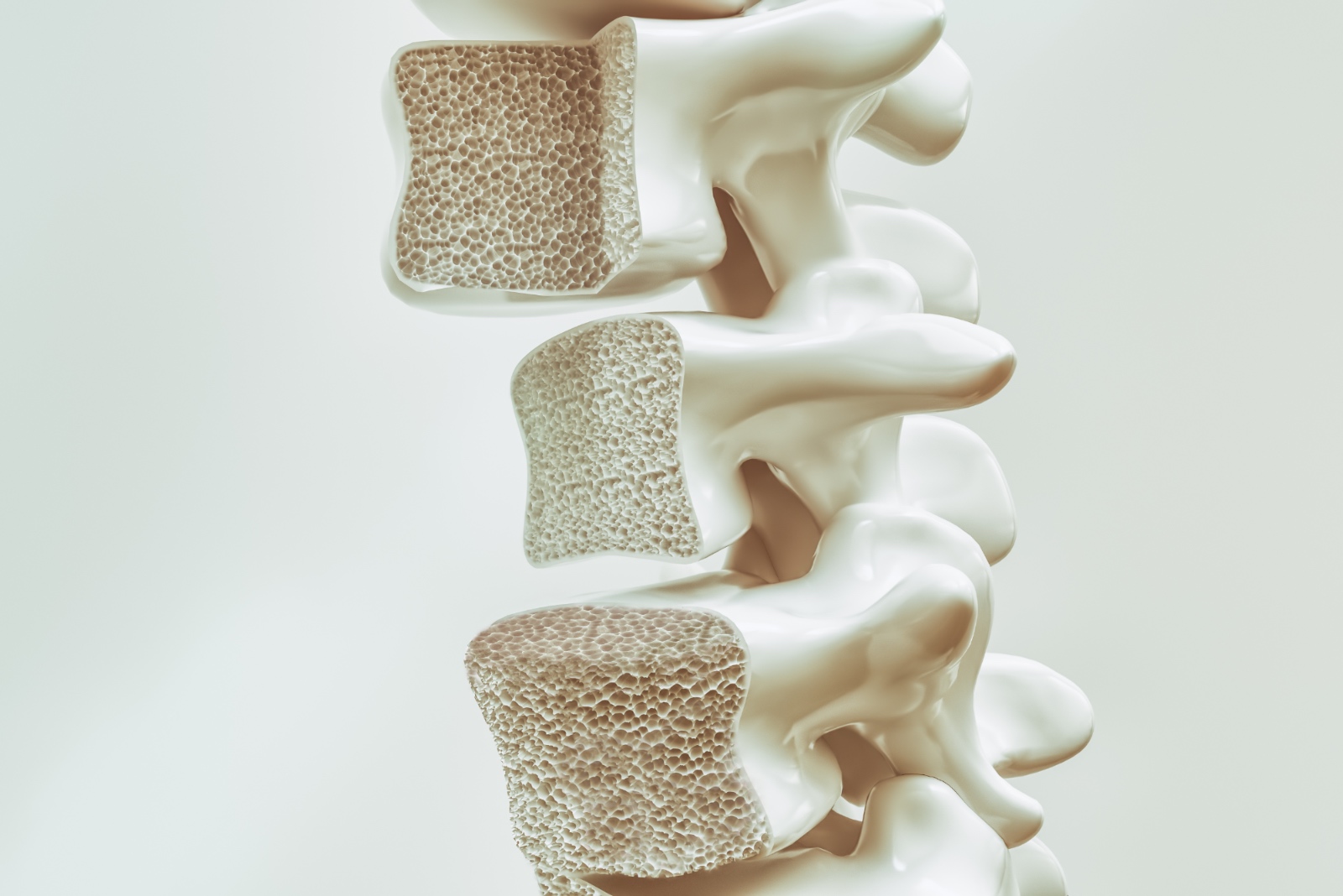
Nature has been highly effective in creating hierarchical structures that are mechanically stable yet contain porosity to serve biological function. This is clearly evident in bone found in the human body. In particular, cancellous bone, which is highly porous, is able to provide mechanical stabilization to the skeleton while supporting the biological function of bone marrow and blood vessels, tissues which are found within porosity of cancellous bone. However, when cancellous bone is used as a bone graft material, the function of the porosity changes. As a graft material, cancellous autograft (bone taken from the patient) or allograft (bone taken from a tissue donor) provide an effective scaffold for new bone formation. Following implantation, new bone and blood vessels will form within the porosity of the cancellous bone structure. In general, this osteoconductive characteristic of bone graft healing is critical to obtaining a successful surgical outcome. The porosity of cancellous bone is one of the primary reasons why autograft is considered the “gold standard” of bone grafting materials.
In the design of a synthetic bone graft material, the more similar a material is to cancellous bone tissue, the more biomimetic (mimicking the natural structure) it is. Although the natural design of cancellous bone is to provide a combination of strength and porosity, it also results in an optimal structure, or scaffold, that allows bone in-growth into the open pores. Due to the importance of porosity on bone graft function, bone grafts that feature biomimetic porosity can have significant benefits for patient outcomes.
An Introduction to Bone Graft Porosity
Porosity is the proportion of empty, connected space within a material. In a bone graft material, this empty space is where new bone formation will occur. In surgical applications where a large void is present in bone, complete regeneration will not take place unless an osteoconductive scaffold is present.
Bone graft porosity plays a key role in the bone formation process, including:
- Providing increased surface area (compared to a solid material) for growth factor and cellular adhesion
- Allowing cellular migration through the bone graft material
- Supporting cellular differentiation and proliferation inside the bone graft material
- Providing a pathway for new vascularization
In short, bone graft porosity is critical to bone healing at a graft site. Although there are some bone graft materials that rely on the spacing between non-porous particles for bone in-growth (e.g. Bioglass and demineralized bone matrix bone grafts), most bone graft materials are porous scaffolds. However, not all pore structures are equal, and very few utilize a biomimetic porosity.
When evaluating a bone graft product, several pore properties should be considered:
- Pore Size
- Interconnectivity
- Pore Volume and Surface Area
Each of these variables can differ substantially from product to product and warrants investigation in more detail.
Pore Size
The size of a pore refers to the diameter across the open pore space. In bone graft material, pore size is a key characteristic that directly affects bone in-growth into the graft. As a reference, bone cells (osteoblasts) are typically ~10-20µm in size. In bone graft materials, pores that are too small (<100µm) can limit bone ingrowth and revascularization of the graft area, while pores that are too large (>500µm) can slow down the bone formation process (Hulbert 1971). In order to facilitate an optimal bone growth response, pore size ranges should typically be between 300-500µm (Kaplan 2005). This will ensure that new bone and blood vessels can grow throughout the entire graft area.

Pore Interconnectivity
Pore interconnectivity is a measure of how well adjacent pores connect to one another, and the geometry of the connection. In an optimal structure, such as cancellous bone, the pores are 100% interconnected and have a uniform geometry from pore to pore. This natural design results in pore channels that span across the entire material on all sides, allowing new bone and blood vessel growth throughout the entire bone graft structure.
Bone graft materials with limited or sub-optimal interconnectivity are characterized by “dead-end” channels that stop bone in-growth, or connection geometries that are highly variable in shape and size. Even when materials have pores in the optimal 300-500µm size range, if the connection areas are smaller, this can create “bottle-neck” porosity. The frequent change from large to small pore sizes can lead to limited bone ingrowth.
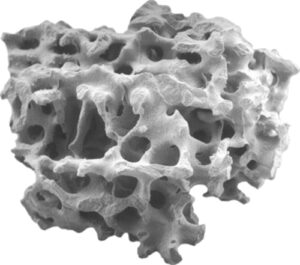
Pore Volume and Surface Area
In addition to pore size, the amount of pores within a material (i.e. the total pore volume) and the corresponding surface area are also important features in a porous graft material. The total pore volume represents the total open space that is available for bone ingrowth. Similar to pore size, bone formation is limited within materials that have low pore volumes. While materials with a high pore volume may seem advantageous by allowing for more bone growth, these materials can become excessively fragile as the empty space volume increases and the material volume decreases. This can lead to crushing of the bone graft scaffold during implantation and elimination of its ability to support bone ingrowth. The increase in brittleness with increasing porosity is due to an inverse relationship between strength and porosity
Also linked to pore volume, the surface area plays an important role in the bone formation response. When a porous bone graft is implanted, the pore surface supports critical elements of the bone regeneration process such as protein adsorption, cellular attachment and proliferation, and cell differentiation. However, not all increases to surface area lead to an improved ability of the graft to support additional bone formation. For example, while increased surface area due to nano-sized surface features can be advantageous for its positive effects on cells and increased protein absorption, the extra surface area from surface morphology does not increase the area available for bone formation. When an osteoblast or stem cell attaches to the graft surface, the surface area of the region under the cell cannot be changed to make more cells attach to that same area. Therefore, in order to increase the osteoconductive function of a graft material, the increase in surface area must correspond to an increase to total pore volume (e.g. more surface area is the result of more pores within a material). While some bone graft products are promoted for their high surface area, this does not necessarily mean that more bone will grow on the surface.
Both total pore volume and surface area represent osteoconductive properties of a bone graft scaffold that can impact bone formation process. However, these characteristics are functionally linked to pore size and interconnectivity. If pores are too small or have limited interconnectivity, bone will not form in these areas. A graft with small pores that prevent bone ingrowth may have a high total pore volume and surface area. However, its function as an osteoconductive scaffold would be limited. Without the ability to support bone ingrowth, these small pores weaken the bone graft’s structure with limited benefit to the healing process.
Advantages of Biomimetic Porosity in Bone Grafts
Although the cancellous bone structure evolved out of a need to provide a mechanical and biological function, it also serves as an optimal blueprint for a porous bone graft material. With uniform pore shape and size, 100% interconnectivity, and sufficient pore volume, this structure provides an excellent osteoconductive scaffold for bone regeneration.
Advanced bone graft materials that utilize a biomimetic porosity, such as Biogennix TrelCor® technology, possess these key features. As seen in the figure below, the structure of TrelCor material is nearly identical to cancellous bone.
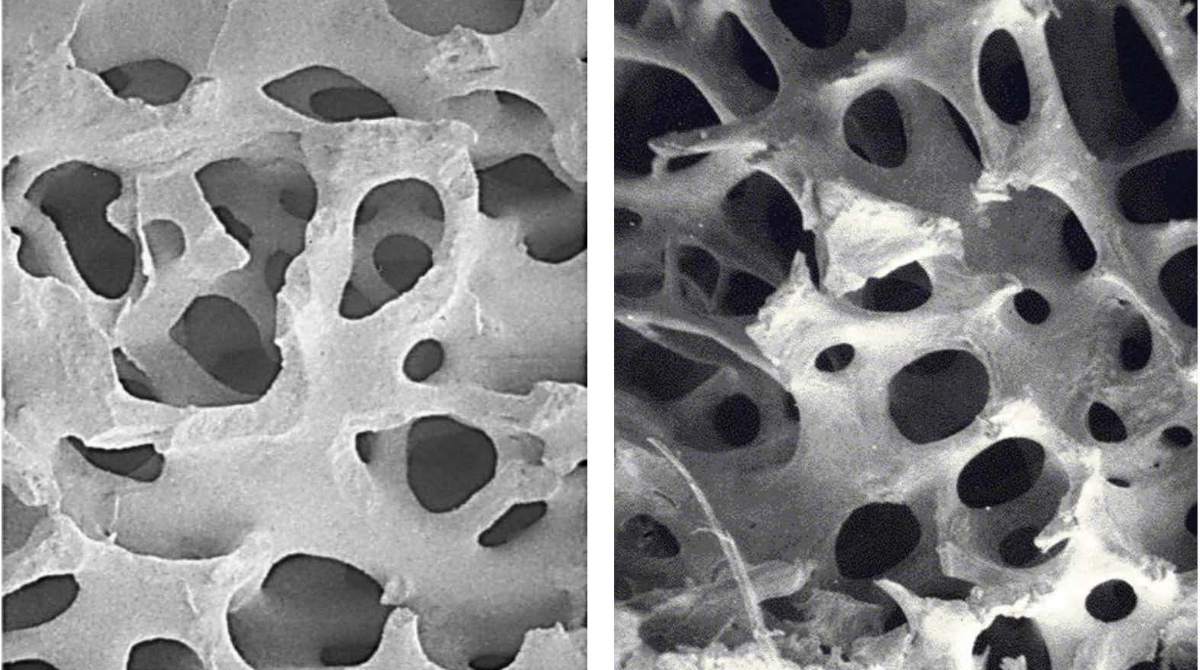
Characterized by an average pore size of 500µm, a 100% interconnected porosity, and uniform pore geometries, Biogennix’ TrelCor material functions as an effective osteoconductive scaffold that can support bone growth throughout the implant material due to its biomimetic porosity. Additionally, this structure results in an optimal combination of strength and porosity that can prevent intraoperative issues arising from graft crushing and brittleness.
Given the importance of porosity on a bone graft’s ability to function as an osteoconductive scaffold, surgeons should select synthetic graft materials that are optimally biomimetic. Preferred synthetic grafts are those that have a porosity that allows for rapid bone formation throughout the entire graft.
As a recognized leader in advanced bone graft technologies, Biogennix is committed to bringing high-quality educational content to our field. This blog will cover technical topics ranging from basic bone graft science to advanced osteobiologic principles. We’ll also discuss market trends and industry challenges. We thank you for reading and invite you to learn more about us here.