Effect of Surface Morphology on Bone Formation

Since their introduction to the market in the 1990s, synthetic bone graft materials have been continually improving and evolving. Over the years, bone graft scientists and engineers have discovered novel materials and manufacturing techniques that can enhance bone formation and improve patient outcomes. As a result of this work, several key characteristics known to impact bone formation have been discovered. This includes:
- Surface morphology
- Surface composition, also referred to as surface chemistry
- Porosity
- Mechanical strength
- Intraoperative handling
This article focuses on the important relationship between bone graft surface morphology and bone formation.
Background: Surface Morphology
One of the primary functions of a bone graft material is to support bone formation on the surface of the implant. This property is called osteoconductivity and is based on the ability of the graft to function as a scaffold for bone growth. Early development of synthetic bone graft materials focused on the use of calcium phosphate ceramics (hydroxyapatite – HA, and tricalcium phosphate – TCP) and resorbable glass (Bioglass). These materials are typically manufactured using a high-temperature process that melts or sinters the material and results in a relatively smooth surface.
Bone graft surface morphology describes the shape, size, and structure of a material’s surface. First-generation synthetic bone graft materials were typically manufactured with a thermal process (i.e. sintering above 1000° C). These high temperatures resulted in graft surfaces that were relatively smooth or contained some micro-features. While these types of surface morphology can effectively support bone formation, they result in a passive bone formation process. This means that the surface morphology and chemistry is biocompatible and bone adheres to it, but it is not actively involved in the cellular bone formation process.
Recent studies have shown that bone graft surface morphology can be improved to actively impact the bone formation response (Zhang, 2017; Duan, 2018). This is accomplished by incorporating nanosized surface features that are typically 200X smaller than a bone cell. This enables a synthetic bone graft material to improve cell function and allow for the differentiation of stem cells.
Types of Synthetic Bone Graft Surfaces
The surface of a synthetic bone graft is controlled by how the materials are synthesized and how the bone graft particles are manufactured. Because porosity is essential for bone ingrowth, the manufacturing process of most synthetic materials is focused on creating a porous scaffold. The resulting surface morphology is usually not considered, or it is a secondary property.
By contrast, newer next-generation synthetic grafts are deliberately processed to create surface morphology and features that can influence the cellular bone formation response.
A variety of manufacturing techniques have been developed over the years and result in varied material surfaces. These surfaces can be grouped into three main categories.
[Note: In the table below, the diameter of a single bone cell (10μm or 10,000nm) is used as a reference.]
| Surface Type | Image |
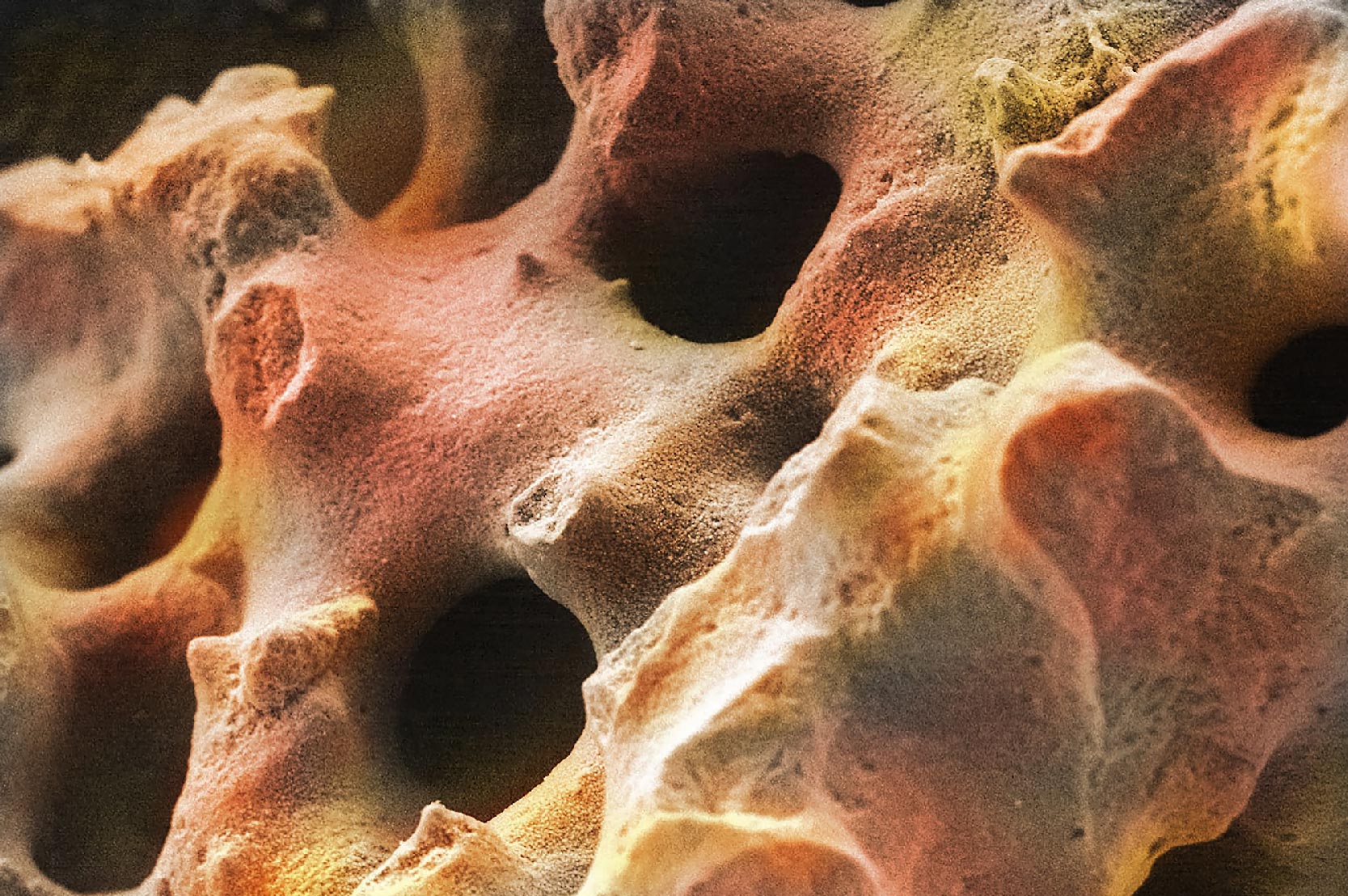
| 1. Smooth Surfaces: These graft materials (such as Bioglass) have a relatively smooth surface and have minimal (if any) micro- or nano-sized surface features. Bone cells attaching to a smooth surface will flatten and spread out over the smooth surface. |  |
| 2. Micro-sized Surfaces: Micro-sized surfaces have features in the micron scale (typically 1μm-500μm). At low magnifications, these surfaces may appear smooth, but moderate magnification (500X) shows the micro-sized structures on the surface. Bone cells typically attach and grow around these surface features. | 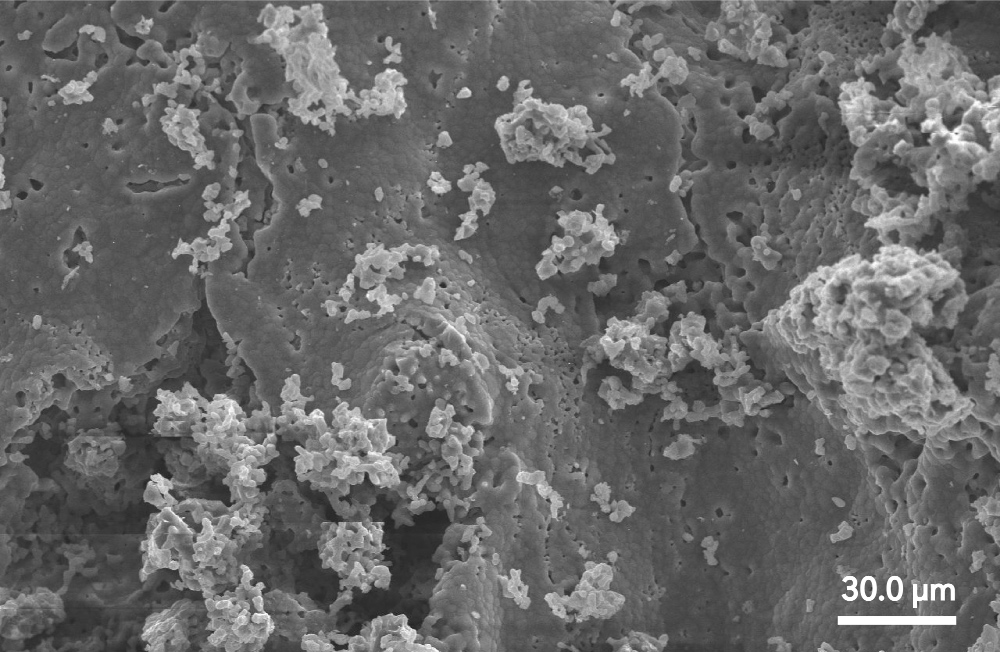 |
| 3. Nano-sized Surfaces: Nano-sized surfaces have features much smaller (<100nm) than a bone cell. High magnification imaging (e.g. 20,000X) is required to visualize nano-sized features. Cells will attach to a region that consists of a significant number of nano-sized features under one cell. |  |
The Influence of Surface Morphology on Bone Formation
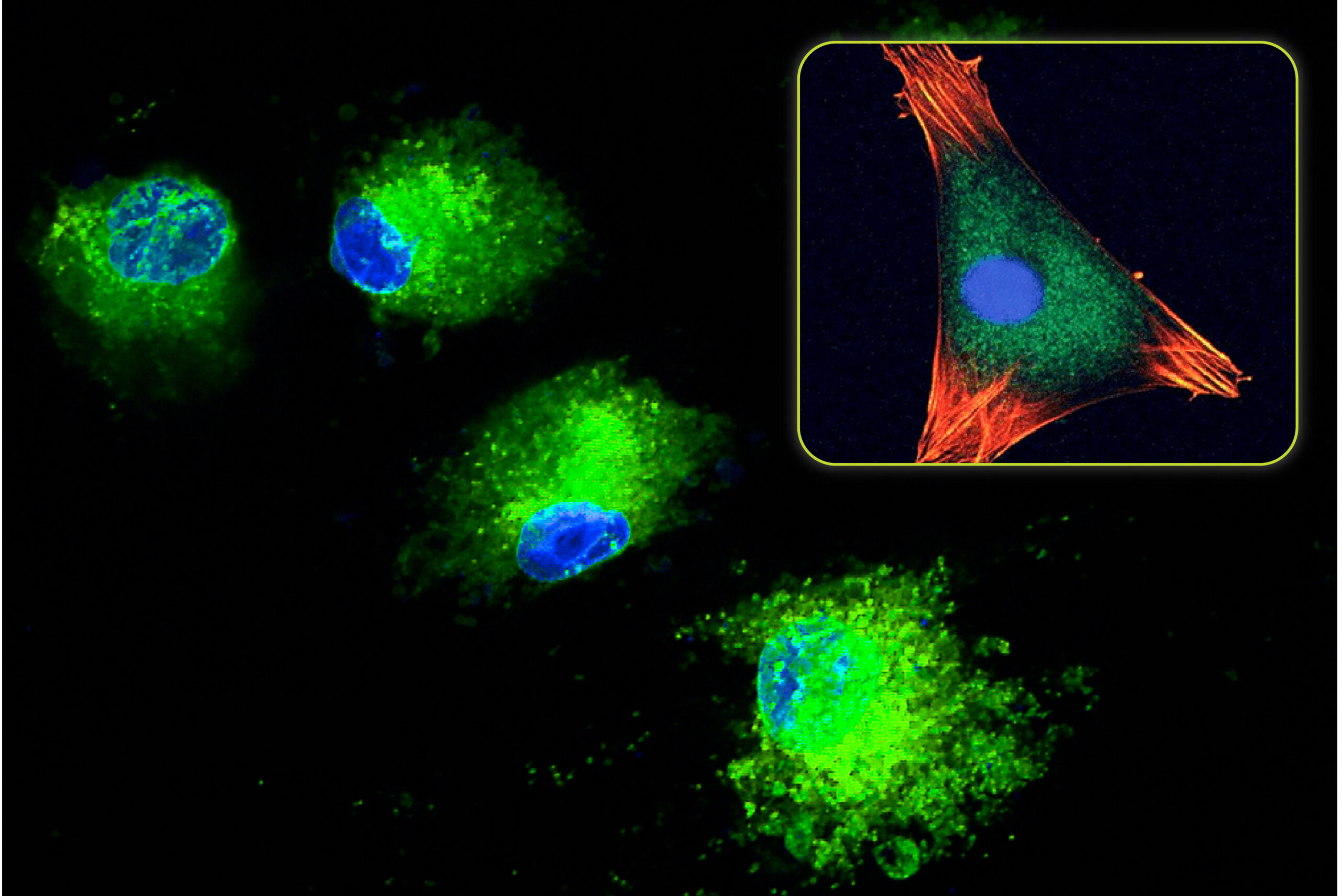
In recent years, scientists have studied the effect of nano-sized surface morphology on the bone formation process. The data shows that a nano-structured surface can positively influence osteoblast function as seen by an increase in cell proliferation and increases to mineralization and protein expression. Additionally, nanostructured surfaces can cause a stem cell (e.g. bone marrow cell) to become, or differentiate into, an osteoblast.
A nanostructured bone graft surface can accomplish this through multiple mechanisms. These properties are dependent on both the nano-sized features and surface composition.
- Mechanotransduction: As osteoblasts attach to a nanostructured surface, the cell membrane is stretched due to the nanosized surface features. This simulates mechanical loading on the cell. Since bone cells positively respond to mechanical stimuli (i.e., Wolf’s Law) (Liu, 2020), the bone formation process is activated by the nanostructured surface.
- Ion Release: When a synthetic bone graft surface is completely covered by nanosized features, the surface area is significantly increased. This leads to faster dissolution of the surface layer (Xu, 2007). One theory to explain this observation is that the ions released from the surface can cause a biologic effect, e.g., the ions increase osteoblast function and cause stem cell differentiation.
- Protein Absorption: The high surface area of a nanostructured material also provides a large area for protein absorption. In general, proteins have a high affinity for attaching to calcium phosphate ceramics (Zhou, 2018). With a nanostructured bone graft based on calcium and phosphate, certain proteins associated with the bone regeneration process can attach to the surface and positively influence the cellular response.
Nanocrystalline Hydroxycarbanoapatite: An Optimal Nanostructured Surface
Nanostructured bone graft materials are typically associated with ceramics. One material of particular interest is nanocrystalline hydroxycarbanoapatite (HCA). Nanocrystalline HCA features a nanocrystalline surface morphology with a carbonate apatite surface composition that allows the material to actively participate in the cellular bone formation response.

Nanocrystalline HCA is found in all Biogennix bone graft products and is a core feature of Biogennix’s TrelCor®technology. The entire surface of the TrelCor material is covered with nanocrystalline HCA. Bone graft products with advanced material surfaces, like the nanocrystalline surface morphology of TrelCor, are rapidly being adopted by surgeons because they can positively influence cellular bone formation and can lead to better patient outcomes.
As a recognized leader in advanced bone graft technologies, Biogennix is committed to bringing high-quality educational content to our field. This blog will cover technical topics ranging from basic bone graft science to advanced osteobiologic principles. We’ll also discuss market trends and industry challenges. We thank you for reading and invite you to learn more about us here.